|
The products from these two pathways are CO 2 and the reduced compounds NAD(P)H 2 and FADH 2, which in turn are used for oxidative phosphorylation (OXPHOS), transferring their electrons to the terminal oxidase where O 2 acts as the final electron acceptor, producing high-energy phosphate bonds (ATP) (Millar et al., 2011 van Dongen et al., 2011). 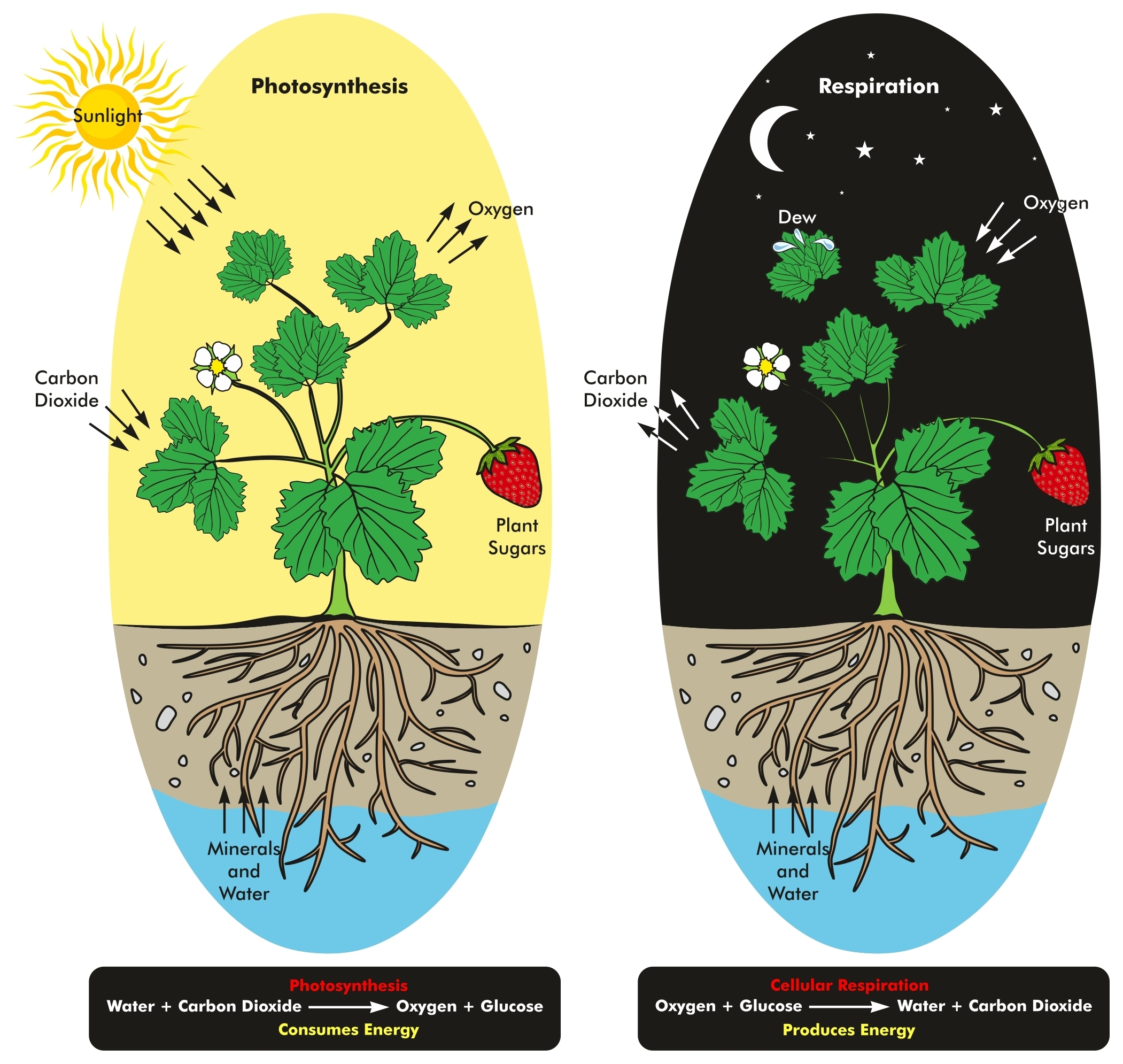
This process oxidizes carbohydrates through two principal pathways: glycolysis and the tricarboxylic acid (TCA) cycle. In plants glucose is the main substrate for respiration. Plants are autotrophic organisms able to use solar radiation to split water molecules (H 2O) and reduce the carbon dioxide (CO 2) compounds that can finally be stored as insoluble polysaccharides (starch) or used directly in the synthesis of other compounds. Key words: Electron transport chain, hypoxia, Krebs cycle, maintenance respiration. This review analyzes the current knowledge on the metabolic and functional aspects of plant respiration, its components and its response to environmental changes. Changes in these components have been reported when plants are subjected to stress, such as oxygen deficiency. To understand the energetic link between these alternative pathways, it is important to know the growth, maintenance, and ion uptake components of the respiration in plants. In this way, in order to avoid states of low energy availability, plants exhibit great flexibility to bypass conventional steps of glycolysis, TCA cycle, and OXPHOS. For example in plants, because their high plasticity, respiration involves metabolic pathways with unique characteristics. Respiratory metabolism is ubiquitous in all organisms, but with differences among each other. Respiration is an oxidative process controlled by three pathways: glycolysis, the tricarboxylic acid (TCA) cycle, and oxidative phosphorylation (OXPHOS). *Corresponding author ( de Investigaciones Agropecuarias, INIA La Platina, Santa Rosa 11610, La Pintana, Santiago, Chile. You should check that out.1Centro de Estudios Avanzados en Fruticultura (CEAF), Camino Las Parcelas 882, sector Los Choapinos, Rengo, Chile. If you still don't understand, the National Library of Medicine has a detailed (albeit complicated) article on anaerobes. This is an extremely simplified explanation (I left out the chemical equations and molecules required for the process as they're not particularly well-known), but I hope this helps! Obligate anaerobes do not have these detoxifiers, and as a result, the toxic molecules interfere with cellular processes. So why can aerobes survive? They have in them specific molecules that react with the dangerous molecules to make something much less dangerous (these detoxifying molecules are superoxide dismutase, catalase, and peroxidase). This extreme reactivity basically tears the cell apart if these new molecules and ions are not dealt with. 
To make a long complicated process short (and simplified), there are certain oxygen products that are produced by processes in the cell that are extremely reactive. Obligate anaerobes have not developed these defenses. 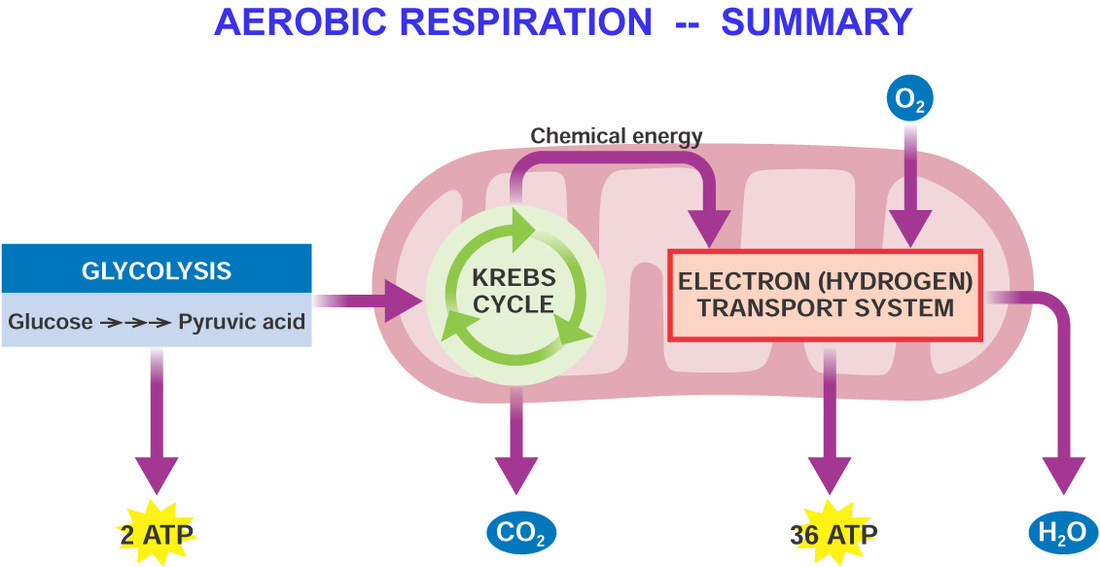
A more electronegative element wouldn't necessarily have any effect on the rate of electron flow down the ETC and therefore wouldn't affect the rate of ATP production.īasically, for aerobic organisms to survive in oxygen, they require "defenses". P.S remember oxygen is not producing the ATP itself it is merely keeping the transport chain unblocked so the electrons keep flowing. Please bear in mind these are just my thoughts. Finally fluoride is known to be damaging to the body above certain concentrations affecting things like the nervous system and hormone secretion as well as protein synthesis. Also if fluorine were used as the terminal electron acceptor it would form HF, hydrofluoric acid in solution which is hard for the cells to deal with and would affect pH in the cytosol affecting enzyme function whereas oxygen just forms water. In addition fluorine is very reactive so would not exist by itself for very long. Oxygen makes up 21% of our atmosphere and is stable in both air and water whereas fluorine is much rarer. The first is simply to do with availability. There are a few reasons that spring to mind.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
